Share Abstract
Inclusion of pregnant and lactating people in the PURPOSE 1 study: efficacy, safety, and pharmacokinetics
Abstract Content:
BACKGROUND: Pregnant and lactating people (PLP) are disproportionately vulnerable to HIV-1 acquisition but historically excluded from Phase 3 HIV trials. PURPOSE 1 (NCT04994509) was the first pre-exposure prophylaxis (PrEP) trial to intentionally include PLP to address their urgent unmet need for HIV prevention options.
METHODS: We engaged community stakeholders, regulatory agencies, ethics committees, and maternal/pediatric health experts to responsibly include PLP in PURPOSE 1. To respect autonomy and reproductive choice, contraception was offered but not required; participants who became pregnant could remain on study drug following additional informed consent. We describe pregnancy outcomes, adverse events (AEs), and HIV infections in PLP randomized to twice-yearly subcutaneous lenacapavir up to the primary analysis. Lenacapavir plasma concentrations in PLP during each trimester/postpartum were compared with non-PLP using a population pharmacokinetics (popPK) model. Lenacapavir concentrations in breastmilk and infant plasma were measured (smaller subset).
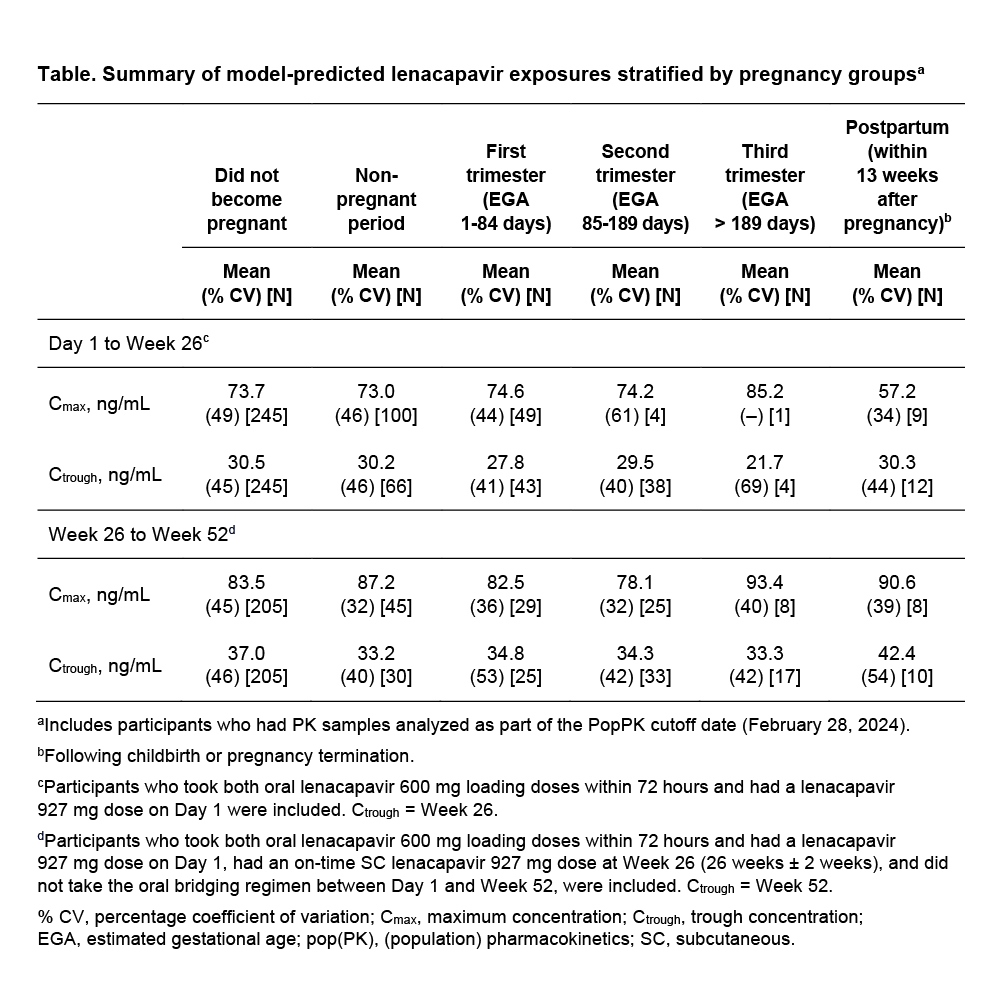
RESULTS: Of 2140 participants receiving lenacapavir, 184 participants had 193 pregnancies, of which 88 (45.6%) were ongoing. The 105 pregnancies with outcomes included 52 live births (49.5%) and 53 losses (50.5%), including 30 induced/elective abortions (28.6%), 20 spontaneous abortions (19.0%), and 3 stillbirths (2.9%). Maternal pregnancy-associated AEs were uncommon, with gestational hypertension/pre-eclampsia (n=4) and hyperemesis gravidarum (n=3) most reported. No HIV infections occurred in PLP receiving lenacapavir. In the PopPK analysis, predicted lenacapavir exposure was not statistically significantly different by pregnancy trimester or postpartum status compared with non-PLP (Table). Lenacapavir was present in breastmilk (median milk-to-plasma ratio: 0.63 [n=8 matched pairs]); however, lenacapavir exposure in infant plasma was minimal (median breastfed-infant-to-mother plasma ratio: 0.05 [n=11 matched pairs]).
CONCLUSIONS: Lenacapavir was efficacious, safe, and well tolerated, with no clinically significant exposure differences in PLP and minimal exposure in breastfed infants. Proactive evaluation of lenacapavir efficacy, safety, and PK data in PLP can support accelerated access to lenacapavir for PLP who need or want PrEP.
METHODS: We engaged community stakeholders, regulatory agencies, ethics committees, and maternal/pediatric health experts to responsibly include PLP in PURPOSE 1. To respect autonomy and reproductive choice, contraception was offered but not required; participants who became pregnant could remain on study drug following additional informed consent. We describe pregnancy outcomes, adverse events (AEs), and HIV infections in PLP randomized to twice-yearly subcutaneous lenacapavir up to the primary analysis. Lenacapavir plasma concentrations in PLP during each trimester/postpartum were compared with non-PLP using a population pharmacokinetics (popPK) model. Lenacapavir concentrations in breastmilk and infant plasma were measured (smaller subset).
RESULTS: Of 2140 participants receiving lenacapavir, 184 participants had 193 pregnancies, of which 88 (45.6%) were ongoing. The 105 pregnancies with outcomes included 52 live births (49.5%) and 53 losses (50.5%), including 30 induced/elective abortions (28.6%), 20 spontaneous abortions (19.0%), and 3 stillbirths (2.9%). Maternal pregnancy-associated AEs were uncommon, with gestational hypertension/pre-eclampsia (n=4) and hyperemesis gravidarum (n=3) most reported. No HIV infections occurred in PLP receiving lenacapavir. In the PopPK analysis, predicted lenacapavir exposure was not statistically significantly different by pregnancy trimester or postpartum status compared with non-PLP (Table). Lenacapavir was present in breastmilk (median milk-to-plasma ratio: 0.63 [n=8 matched pairs]); however, lenacapavir exposure in infant plasma was minimal (median breastfed-infant-to-mother plasma ratio: 0.05 [n=11 matched pairs]).
CONCLUSIONS: Lenacapavir was efficacious, safe, and well tolerated, with no clinically significant exposure differences in PLP and minimal exposure in breastfed infants. Proactive evaluation of lenacapavir efficacy, safety, and PK data in PLP can support accelerated access to lenacapavir for PLP who need or want PrEP.
